This is short post, just to let readers know I have to take a break to recover my hands, and others may write in my stead. Hope to come back soon!

This is short post, just to let readers know I have to take a break to recover my hands, and others may write in my stead. Hope to come back soon!


It’s a fact that most people with HNPP know, but there are suggestions that certain exercises can actually do more damage than good. From walking to some types of yoga, while most forms of physical activity are encouraged by gym instructors, our bodies tend to rebel from the norm. But can exercise actually induce HNPP, even though it’s generally thought to be an hereditary condition?
As HNPP is an autosomal dominant disorder in which the condition is inherited and there is a deletion of one of the genes associated with PMP22, a child of an affected individual is at a 50 per cent risk of being affected by the condition. This suggests that the likelihood of a person developing HNPP out of nowhere is relatively low.
What are ‘sporadic’ cases of HNPP?
There have been recent reports that state that a few types of muscle training has actually brought about sporadic symptoms of HNPP without any previous familial history of the condition. Sporadic cases due to de novo deletion accounted for 21 per cent of the investigated HNPP families, as reported by some studies. De novo mutation is an alteration in a gene that is present for the first time in one family member as a result of a mutation in a germ cell of one of the parents or in the fertilised egg itself.
According to a 2017 study carried out by researchers at the Department of Neurology and Rheumatology, Shinshu University School of Medicine, Matsumoto, Japan, this apparently was the case in a 15-year-old boy. The authors suggest that this is the first instance of an adolescent “that developed neurological symptoms during muscle training in a school baseball club activity”, the first signs in a sporadic case of HNPP. The teenager developed bilateral painless brachial plexopathy through short-term barbell training and plank exercises.
“Patients sometimes show an atypical clinical phenotype, and a diagnosis of HNPP can therefore be challenging, especially in sporadic cases as in our patient.”
“Muscle Training-induced Bilateral Brachial Plexopathy in an Adolescent with Sporadic HNPP” – Kodaira, M., et al – July 2017
While brachial plexopathy can be a common feature of HNPP, bilateral involvement is thought to be unusual because HNPP is usually associated as a mononeuropathic condition, where there is a focus in only one area of nerves.
The authors in this case say: “As his clinical and NCS [nerve conduction study] findings indicated muscle training-induced bilateral brachial plexopathy in HNPP, genetic analysis for this disorder was performed, which revealed deletion of the PMP22 gene. Patients sometimes show an atypical clinical phenotype, and a diagnosis of HNPP can therefore be challenging, especially in sporadic cases as in our patient.”
The major difference in this occasion is that not only did he suffer from a bilateral injury, he also developed a sporadic case of HNPP triggered by relatively straightforward exercises. This is unlike some of the other examples where those in military training are far more likely to develop symptoms linked to HNPP, due to the strenuous physical activity undertaken by soldiers. However, this is not the first time ‘push ups’ are seen to be the main culprit as a trigger.
In another 2017 report A Case of HNPP due to Push-up Exercise, a 17-year-old man with no familial history of the condition developed “motor and sensory disturbance of the left upper limb a few days after starting push-up exercise”. Scientists from the Department of Neurology, Hirosaki University Graduate School of Medicine, say that the patient also developed brachial plexopathy, similar to the case above.
In the abstract, they conclude that: “Genetic tests revealed a diagnosis of hereditary neuropathy with liability to pressure palsies (HNPP). HNPP should be included in the differential diagnosis for neuropathy due to slight exercise or nerve compression even when familial history is negative.”
The original text is in Japanese – read full document here.
Researchers from the Division of Neurology and the Department of Pathology, University of Missouri School of Medicine, Columbia, USA, found a case in 2004 akin to the above studies. A 21-year-old in good health began to develop symptoms of HNPP on her first day of military training. She began to show symptoms of severe pain, weakness, and atrophy in her right shoulder, foot and hands. Her mother and her family had no history of neuromuscular disease. She did not know her father or his family history.
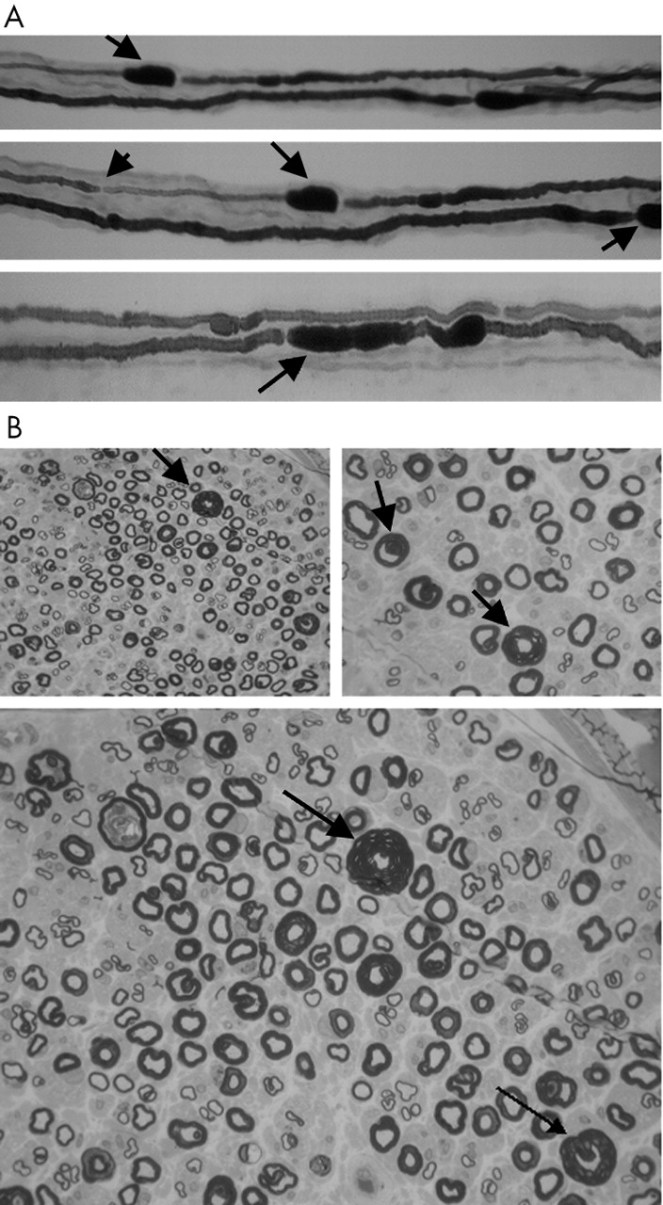
Researchers of the study Fulminant Development with Axonal Loss during Military Training say that the patient’s symptoms were “unique” and that she did not have the “cardinal features of HNPP” including inheritance and mononeuropathy.
They say: “While the presentation and severity of this patient’s condition may relate to a specific unknown genetic profile with very low PMP-22 mRNA levels, Schenone et al, in finding correlations between reduced PMP-22 mRNA levels and disease severity, also suggested that extrinsic factors—for example, level of physical activity, may be important in determining phenotypic features.
“This appears to be true of our patient—that she was neurologically normal, then developed symptoms on the first day of military physical training with progression as she continued the training over a three week period, suggests that disease severity and focal axonal damage were related to these intense activities.”
The authors add: “Additionally, as it has recently been recognised that sporadic cases of HNPP are common, either because of de novo mutations or asymptomatic carriers, her lack of family history did not preclude this diagnosis.”
How likely is it that a case of HNPP is sporadic?
It’s important to note that HNPP may not be easily traced in other family members, which means while it may appear sporadic, it could just have been overlooked. A 2013 case report reiterates this view, with authors saying that approximately one-third of deletion carriers are unambiguously detected on the basis of “electrophysiological criteria and confirmed by genetic analysis are asymptomatic and do not display significant signs at clinical examination.”
Researchers from Department of Paediatrics, Hospital de Guimarães, Portugal, say: “Thus, the family history is often uninformative, and a significant proportion of probands may be considered as apparently sporadic cases. However, a close questioning and examination of the relatives provided evidence for autosomal dominant inheritance in families that were originally stated by the probands to be normal. Therefore, HNPP can easily be overlooked in those cases in which familial involvement is not recognised unless intensive ascertainment techniques are used.”
Electrophysiologic studies are said to be “suggestive” and not “sufficient for diagnosis” which may be why it may be imperceptible to locate.
How exercise can trigger symptoms
The more frequent representations are in those who already have familial histories of HNPP, or in those who have already been diagnosed. In one such case in 2005, hiking and athletic training brought on the symptoms. A 10-year-old girl, who suffered from acute, recurrent monoplegic episodes affecting both the sciatic nerves and the left brachial
plexus since the age of 7, showed quite a lot extensive symptoms from relatively low impact exercise.

Authors from the Department of Paediatrics, Yokohama City University Medical Centre, Japan, say: “This school child having HNPP is considered to be susceptible to the influence of abundant physical training, rather than minor trauma or compression at sites of entrapment of peripheral nerves.” However, this is hardly surprising given the nature of the condition. What’s more unusual are the situations where HNPP is not present beforehand or without familial background.
It goes without saying, exercise can cause many types of injuries even for those without HNPP depending on how extreme it may be. And as we have seen above, there have been at least several cases of ‘sporadic’ HNPP, which develops without any prior history of symptoms or familial connection.
However, it may be worth noting that without the correct tests to detect hereditary links, these may be incorrectly termed as ‘sporadic’. That being said, there have been established reports that suggest at least 21 per cent of HNPP cases are de novo mutations, so it may not be out of the realms of possibility that certain types of exercise can induce symptoms of HNPP.

Having HNPP thankfully isn’t the end of the world, but it does make things rather difficult when it comes to relationships. This may not apply to you if like many of the individuals on the HNPP Facebook groups, are already happily or regularly married or in a relationship of any kind. But for those who are trying to balance both worlds, many become apprehensive of making another commitment, when all you’re trying to do is look after yourself. So how do you manage?
Relationships are always work. Add significant stress to the situation and they’re extra work. And HNPP falls under the “stress” umbrella, which tends to add complications to every stage of a relationship, just as it gets in the way of everything else.
Dr. Morwenna Opie, a Clinical Psychologist who works at the Nightingale Hospital in London, UK, says that “healthy” relationships are important. Dr. Opie, who has been diagnosed with POTS secondary to Sjögren’s Syndrome, states: “Healthy, supportive, and fun relationships can be our greatest asset in shaping a happy and fulfilling life, and this is especially the case when opportunities for physical activity are more limited.”
However, she adds a warning to this statement: “maintaining unhealthy relationships can be more toxic to our health as those chocolate binges or caffeine or whatever else we might have resolved to do away with this month. The evidence continues to accrue demonstrating that social stressors and anxiety takes their toll on our immunological functioning, and all aspects of our physiology, with the potential to cause a vicious cycle of deteriorating physical and mental health – and relationships.”
https://twitter.com/ClarkDeHart/status/886353231525425152
The most important relationships she says are friendships which require constant maintenance just like any other kind of connections, which is the first step ahead of dating. Think of it as the steps to rehabilitation. Unlike trying to stay sober, your priority is first to yourself and maintaining the connections you already have. Hence, just like in Alcoholics Anonymous, it is recommended that people who are still within the first year of their recovery should avoid beginning romantic relationships.
The first few months of recovery are often described as an emotional rollercoaster because there is so much going on. The last thing that an individual will want to do will be to add the stress of a new relationship to the mix. It is going to take all your attention to make it through this early part of recovery. It’s also important for you to come to terms with yourself during this time.
As a result, the worry for many trying to date for the first time after a diagnosis is seeing yourself differently, acceptance from the other person, and generally managing the symptoms on a day-to-day basis. Therefore, there are several questions you need to ask before making that leap.
Are you in a position to date?
What to look for in a partner
You’re ready to jump into the crazy world of dating, so now what? Well, before filling up your calendar with potential male or female suitors, you should have a clear idea of the type of qualities to look for in a mate. Best to look for a partner and not a caregiver:
Your date should be:
When should you reveal your condition?
Dr. Gail Saltz, M.D., a renowned psychoanalyst, columnist, bestselling author, says: “In the dating world, it’s really about when you choose to discuss the topic of illness. It’s important to be thoughtful about when might be the best time; not disclosing this part of yourself too early or waiting too long.”
One of the biggest challenges about dating when you have a chronic illness is trying to figure out when to tell the person you’re dating about your disease. Some people will tell you that you have to wait until things are more serious between you two before the big reveal about your illness.
https://twitter.com/Sarafina321/status/861255355631886340
Other people will tell you it’s absolutely mandatory to inform them up front, because they should understand that dating you might have some challenges so they can decide if those challenges outweigh the awesome privilege of getting to be with you. There is no exact science to it.
The most important part is to not feel obligated to share such a sensitive and personal part of your life if you’re not ready yet. Let your illness come up naturally (well, as natural as a discussion about a chronic illness can be anyway) and when you feel comfortable.
Ken Robbins, MD, a clinical professor of psychiatry at the University of Wisconsin–Madison says that if you’re especially worried that your health secret “is likely to define you before the person has gotten a chance to know you at all”, then don’t mention it on the first date.
“I don’t talk about my illness on a first date. I may mention why I don’t eat gluten or, “Yeah, I have a bum knee so I can’t run! It sucks!” But I don’t dive into details.”
“How I Learned to Date With a Chronic Illness” – Jacqueline Raposo, Cosmopolitan
But there’s one exception and that’s if personal information about you living with a chronic illness is already out on the internet. In this case, you may want to tell your date sooner than later because there’s a good chance they have Googled your name and found out about you.
How should you reveal it?
Unfortunately, there’s no easy way to tell your date about your chronic illness. Here are some tips to revealing information:
Dating with chronic illness is hard for sure, and there are times when you may feel undateable. Self-care should still be your number-one priority. But there are many things you can offer your dates because you’re much more than your illness.
You could be a great listener, a deep empath, an entertaining storyteller. Sometimes dating is a great way to get yourself out of your head and out of bed, even when the latter seems impossible or undesirable at best.
In the words of Deepak Chopra:
“To value yourself is to love yourself. It is really from here that your love for others comes. If you value yourself a great deal, you actually have something to give to others.”
The fastest way to love and connection is to show the other person who you are right now, in this imperfect moment.

There can be a lot of worry and anxiety that arises during pregnancy, one of which includes what to expect, especially with HNPP. Many can have a more or less seamless experience while others find that their symptoms are exacerbated during this time. So what is the ‘norm’ of HNPP during pregnancy?
“In my 30’s during pregnancy, I had sciatica because I had enormous babies resting on my spine. Bladder too, but peeing my pants when I sneezed or laughed – seemed like something that just happens to pregnant women.”
A mother with HNPP from the blog Chronic Pain Journal
First of all, HNPP does not affect the fetus or the pregnancy itself thankfully. However, during pregnancy, symptoms that manifest due to HNPP such as palsies, sciatica, or pain in the lumbar region, may be heightened as a result of added pressure on the body.
Disclaimer: Please ask your medical practitioner for more information. This article is based on various research, journals and testimonies.
How does HNPP manifest during pregnancy?
According to Dr. Rakesh B Vadhera, an obstetrics anaesthesiology consultant and professor at the University of Texas, alongside Dr. Michelle Simon, a paediatrician and neuropathology expert, peripheral entrapment neuropathies are common during pregnancy and may lead to “severe discomfort”.
Writing in the book Maternal Medicine published in 2015, Dr. Vadhera and Dr. Simon state: “Pregnancy itself may predispose patients to some of these entrapment neuropathies, which are mostly benign in their evolution and prognosis and will resolve spontaneously in the postpartum period.” This appears to be good news for expecting mothers concerned that the symptoms may not disappear after the birth of the child. For all that however, there have been cases of symptoms lingering postpartum.
They add: “Delivery may predispose patients to compression or stretching of some nerves and plexuses that may precipitate symptoms. Prompt clinical evaluation and, when necessary, an electrophysiologic evaluation may aid in the diagnosis and subsequent management.” As briefly mentioned in the article Is surgery worth it with HNPP?, it’s vital to let your medical team know how to make you comfortable during this time as well as through labour, to avoid further nerve-related damage. This is addressed in more detail below.
What symptoms to expect when you’re expecting
In some extreme cases of Charcot Marie-tooth-related disorders, the obstetricians above say pregnancy can affect respiratory muscles and thoracic vertebral anatomy, “impacting patient respiratory function during pregnancy and affecting delivery and anesthetic care”. But this may be evident during the third trimester when there is added strain on the body, and therefore you may have enough warning to consult a health professional beforehand.
Author Dr. Pierre Bouche, based in the Department of Clinical Neurophysiology, Salpêtrière Hospital, Paris, France, says that in some neuromuscular disorders, carpel tunnel syndrome (CTS) could also manifest during pregnancy.
In the edition Peripheral Nerve Disorders as part of the Handbook of Clinical Neurology, Dr. Bouche states: “[Carpal tunnel syndrome] can develop at any time in pregnancy, but it is most frequent during the third trimester and may be due to fluid retention exerting pressure on the median nerve.” However, this can vary from person to person depending on how sensitive the nerves are around the wrist and upper arm.
Other areas that may be affected can also differ. Authors of the medical reference guide Obstetric Anesthesia and Uncommon Disorders, 2008, reiterate that HNPP may exacerbate neuropathies associated with pregnancy and delivery. They say HNPP symptoms such as “lumbosacral plexus, femoral, lateral femoral cutaneous, obturator or peroneal nerve palsies” may be aggravated during this time.
But that’s just some of the ways the symptoms may manifest. There are some mothers featured in the Facebook HNPP groups, who have spoken about pain in the ribs, loss of functionality in the legs, arm and leg aches, and the list goes on. On the other hand, there are others who faced symptoms no worse than pre-pregnancy.
How to prepare for labour and delivery
Similar to the diverse responses on how mothers are affected during pregnancy, the same is apparent with the delivery itself. Some mothers elect to have a natural birth, while others require or request cesareans. Using gas, on the other hand, may pose a risk as it is considered a neurotoxin. There have been reports of “heightened pain” with gas according to some users in the HNPP support networks.
“I was 33 when I had the epidural – which triggered my chronic neuropathic pain. The majority of my pain, travelling along the entire right side of my body. Strongest in all the places I had experienced pain during my life. It was like it was the “Red Button” got pushed and a bomb exploded in my Central Nervous System.”
A mother with HNPP from the blog Chronic Pain Journal
According to Dr. Guy Lepski and Dr. J.D. Alderson of the Department of Anaesthesia, Northern General Hospital, Sheffield, UK, dense local anaesthetic blockade should be avoided as it may mask a compression neuropathy. They recommend the following management principles for doctors in the 2001 study Epidural Analgesia in Labour for a Patient with Hereditary Neuropathy with Liability to Pressure Palsies:
Both Dr. Lepski and Dr. Alderson say that the “Labour progressed uneventfully and there were no neurological sequelae following delivery”.
In addition to these suggestions, David H. Chestnut alongside several other authors have written about safeguards to minimise peripheral nerve compression. In Chestnut’s Obstetric Anesthesia: Principles and Practice E-Book, they advise:
A report by French researchers S. Berdai and D. Benhamou from the Department of Anaesthesia and Resuscitation, Bicêtre Hospital, Le Kremlin-Bicêtre, suggests that it is possible to have an epidural as well as spinal anaesthesia during labour. In the report Regional Anaesthesia for Labor and Delivery in a Parturient with Neuropathy with Liability to Pressure Palsy, a woman had two cesarean sections, one with an epidural that resulted in no “neurologic complaints in the postpartum periods”.
They say: “For the first delivery, epidural analgesia was performed for labour pain control but a caesarean section was necessary because of failure to progress (0.0625% bupivacaine with 0,2 μg/ml sufentanil for labour then 2% lidocaine with adrenaline for surgery).
“Two years later, the patient was again seen for a preanaesthetic visit because elective Caesarean section was planned. Spinal anaesthesia using hyperbaric bupivacaine and sufentanil was used. Both deliveries were uneventful”. Uneventful being the operative word.
It is essential to get the right advice while pregnant as well as during childbirth itself, and also on how to manage any symptoms that appear postpartum. Creating a birthing plan will therefore be necessary to avoid any extra issues. That being said, symptoms fluctuate from person to person, which means you may be fortunate enough to have hardly any bumps in the road.
Read: What to avoid with HNPP

When your face unexpectedly becomes numb, or begins to spasm, many HNPP sufferers do wonder what godforsaken cause could be behind it. Yet, it isn’t as uncommon as many may believe. Health professionals say that facial issues tend not to be associated with the condition, hence multitudes have been diagnosed with Bell’s Palsy instead.
What is Bell’s Palsy?
Bell’s palsy, or idiopathic facial paralysis (IFP), is the most common cause of unilateral, lower motor facial palsy. It’s origins remain uncertain. However, the first familial occurrence was found in 1887, hence hereditary factors have been considered to play a role in the etiology of the disease. It is believed to occur when the nerve that controls the muscles in your face becomes compressed and IFP is generally linked to inflammation or viral infections.
“In the last year and a half I have been dealing with one sided facial pain and numbness. It has evolved into a burning that sequentially involves the tongue, then lips, then cheek, then eye, then ear and finally throat all on the left side. It waxes and wanes and seems to do so based on my stress levels. I have had CT and MRI, normal; as well as a battery of blood tests, normal; seen a neurologist who said I’m fine (ahem!) and an ear nose and throat doc who says I have non-motor Bell’s Palsy (my research indicates there is no such thing).”
User on the BrainTalk Communities Forum
It’s obvious that there could be secondary condition causing facial issues, but several members of various HNPP groups have spoken about pain, weakness numbness, and spasms associated with the head and face.
Scientists have been studying the link between IFP and HNPP in order to get a better understanding of both causes. A letter in the Journal of Clinical Neuroscience in 2013 shows how the two conditions are not connected. Researchers at the Department of Neurology, Eginition Hospital, in Athens, Greece, hypothesised that a handful of participants in a study for Bell’s Palsy could have the same mutation for HNPP.
They say: “There are a few case reports of patients belonging to these subcategories of Bell’s palsy, on whom the characteristic deletion of a 1.5-Mb region on chromosome 17q11.2-12 which includes the peripheral myelin protein 22 (PMP22) gene, was detected.”
Out of a 145 unrelated Greek patients with Bell’s palsy, 28 patients with recurrent facial palsy and 18 patients with familial facial palsy were tested for a deletion of the PMP22 gene. However, none of the participants had this mutation.
“These cases could be part of a diverse spectrum of miscellaneous disorders including HNPP.”
“Is there a common genetic background?” Karadima, G. et al, 2013
They conclude: “Bell’s palsy seems to have a different etiology than HNPP. The same applies to familial or recurrent Bell’s palsy. A molecular genetic investigation for HNPP seems to be indicated in cases of recurrent or familial facial palsy accompanied by peripheral nervous system damage or exhibiting a family history of peripheral neuropathy. These cases could be part of a diverse spectrum of miscellaneous disorders including HNPP.”
This is reiterated in the 2009 case report Familial Recurrent Bell’s palsy conducted by researchers from the Department of Neurology, Zhejiang University, Hangzhou, China. Three families in which eight patients had a total of 12 episodes of typical Bell’s palsy were recorded in a bid to find the etiology of the condition.
In spite of this, the researchers infer: “Recurrent facial palsy can occur in neurological disorders such as Melkersson-Rosenthal syndrome (MRS), Moebius syndrome, Charcot-Marie-Tooth disease and hereditary neuropathy with liability to pressure palsy (HNPP). These conditions however, have additional features that distinguish them from idiopathic familials Bell’s palsy. None of our patients have any symptoms indicative of such diseases.”
So what’s the reasons behind facial numbness?
There are several possible causes of facial numbness, also known as hypesthesia. Most of these causes can be traced to a problem in or affecting the trigeminal nerve.
It is one of twelve cranial nerves and is one of the most widely distributed nerves in the head. The cranial nerves can be categorised as two main nerve types: those that control motor responses such as blinking, chewing, or eye muscle movement, and those that respond to the sensations of taste, smell, hearing, and touch.
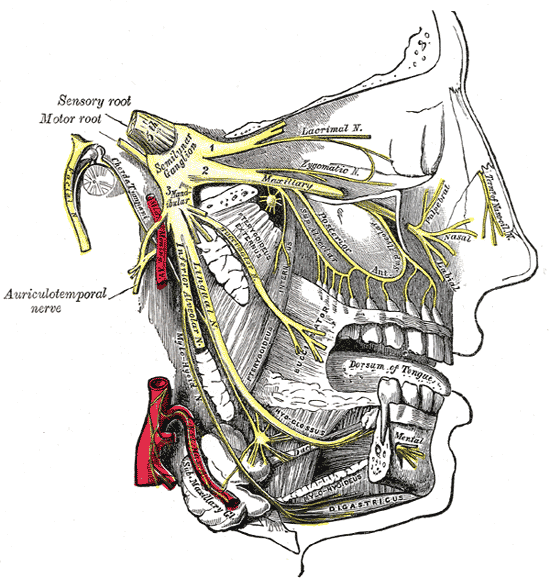
The trigeminal nerve has three branches, which controls both the sense of touch in areas in the face as well as the motor function associated with chewing. Damage to this nerve could, therefore, make chewing difficult, if not impossible. Some sufferers of face numbness also experience numb lips. Or it could create either a ‘pins and needles’ sensation or a loss of feeling in parts of the face. Of the twelve facial nerves, it is usually considered number five. Other parts affected include:
While most of these are connected with the central nervous system, and HNPP is yet to have established links to the CNS, there have been cases where some with the condition have had issues with this particular nerve.
According to a 2015 study carried out by Japanese researchers from Department of Neurology, Osaka Red Cross Hospital, Osaka, there were two cases with cranial involvement without progressive muscular atrophy (PMA). They state: “a 40-year-old female case of HNPP with the involvement of the trigeminal, facial and hypoglossal nerves, and a case of 7-year-old boy having a homozygous deletion of PMP22, who had the LMN [lower motor neuron] impairment in the cranial nerves of VII and III, sensory disturbance in extremities.”
Like many other publications, the researchers state that because of limited studies, “additional investigations are warranted to better understand PMP22 regulation in the CNS and the peripheral nervous system”.
The rarity of such finding is highlighted in a Brazilian study from the Department of Neuroscience at the University of São Paulo. In the 2016 study Clinical and Neurophysiological Features of HNPP, 39 patients were reviewed for neurological symptoms while 33 were given nerve conduction tests. Only one presented cranial nerve related symptoms in terms of “involvement of the trigeminal nerve and other one an episodic involvement of the eyelid branch of the oculomotor nerve”.
They go on to restate: “Cranial nerve involvement was rare in our population […] It seems that this is the pattern in most studies Interestingly, we have previously described a HNPP patient that developed dysphagia. Other rare manifestations in our patients were pes cavus and nerve thickening, as seems to be the case in most studies.”
In the 2006 book Differential Diagnosis in Neurology by Robert J. Schwartzman, MD, the Professor of Neurology documents daily morning reports with neurology residents and the examination of patients in front of colleagues over the last 30 years. The Emeritus Professor of Neurology at the Drexel University College of Medicine in Philadelphia, recounts that “facial nerve involvement occurs rarely” with HNPP.
Therefore, it’s important to realise the symptoms instead of the condition as knowing that it is HNPP, or in this case, may not be HNPP-related, does not change the fact that these symptoms are manifesting.
For many with facial issues, including those suffering from HNPP, it’s vital to get it checked by a medical professional as it could be associated with another underlying or even acute disorder. In many situations, doctors may dismiss it as HNPP and then resort to medication associated with the inherited disorder, because they aren’t certain of what it may be, which is where your persistence to get the correct treatment will be absolutely key.
One of the first responses that seems to appear on search engines about Hereditary Neuropathy with Liability to Pressure Palsies is its relation to the inherited condition Charcot Marie-tooth syndrome (group of disorders). While CMT1A – a subtype of the CMT – is thought to be the most common inherited neuropathic condition, little research is still available to its ‘sister’ condition HNPP. So how do the conditions diverge and converge?
The irony of the conditions being similar in nature, is the fact HNPP is the genetic opposite of CMT1A. HNPP is associated with a deletion on the same chromosomal site where CMT1A (the most common type of CMT) has been found to have a duplication, and it contains an important myelin gene, peripheral myelin protein-22 gene (PMP22).
The chromosome in question is chromosome 17, where CMT1A has an extra copy of PMP22, and HNPP results from the loss of a copy of PMP22. The PMP22 gene encodes for peripheral myelin protein, and disruption of this gene leads to a dysfunctional myelin sheath on nerves.
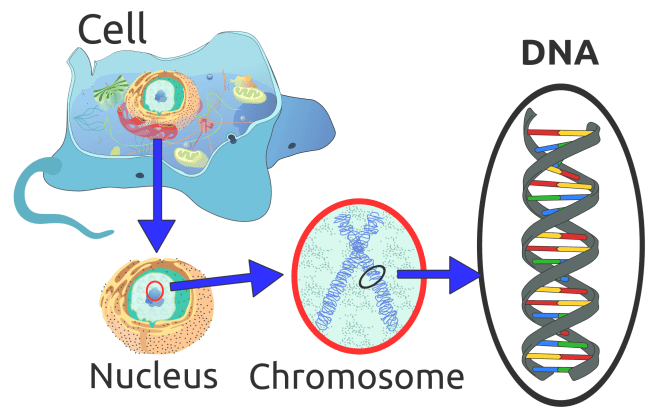
Going back a step and remembering the old science lessons from school, a cell’s nucleus contains chromosomes – rod shaped bodies found in the nucleus of cells that contain genetic information in the form of DNA.
DNA (deoxyribose nucleic acid) is made up of short sections of genes – inherited from parents, and makes up DNA which controls part of a cell’s chemistry, particularly protein production. Each gene codes for a specific protein by specifying the order in which amino acids must be joined together.

In total, humans normally have 46 chromosomes in each cell, divided into 23 pairs. Two copies of chromosome 17, one copy inherited from each parent, form one of the pairs. In one of the arms of the chromosome 17 (if you think about it as an ‘X’ type shape), specifically the ‘P-arm’ lies the gene PMP22 labelled as 17p11.2, which is the area where it resides. The genetic defect in most HNPP patients is a 1.5 Mb deletion (used to describe the length of a DNA/RNA molecule) on this chromosome containing the PMP22 gene.
HNPP is usually caused by an autosomal dominant gene, which means one parent must be affected. While there should be two copies of the PMP22 gene, there usually is only one or some form of mutation, unlike with CMT1A which tends to be a duplication of the gene. As a result, HNPP and CMT tend to be lumped together under one category and while there may be an overlap, there are other forms of CMT and HNPP that do not follow this rule.
The result of this is that the symptoms of each condition resemble one another because they both end up becoming a form of demyelinating neuropathy.
Point mutations in the PMP22 gene are a rare cause of HNPP. In one case, a novel PMP22 splice site mutation – a genetic mutation that inserts, deletes or changes a number of nucleotides in the specific site – was reported in an HNPP family. Nucleotides are organic molecules that form DNA and RNA (ribonucleic acid, which is an important molecule with long chains of nucleotides).
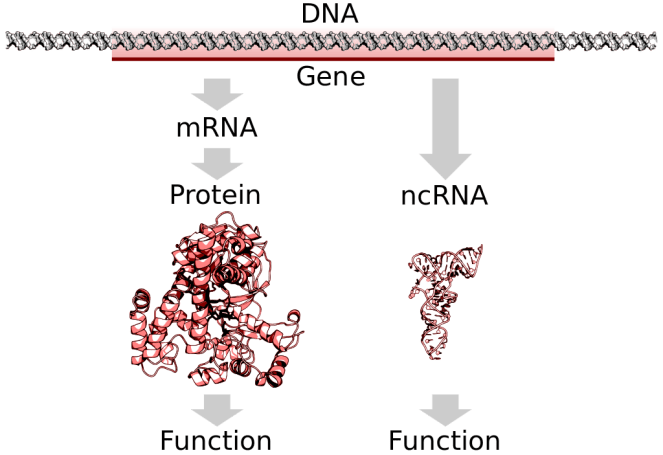
According to a 2006 study, a mutation at nucleotide c.179+1 was found in the PMP22 gene. This mutation causes the synthesis of an abnormal mRNA. Messenger RNA (mRNA), are molecules in cells that carries codes from the DNA in the nucleus to the sites of protein synthesis in the cytoplasm (the ribosomes).
Point mutations of PMP22 gene cause a wide variety of demyelinating neuropathies including HNPP, Charcot‐Marie‐Tooth disease type 1A (CMT1A), Dejerine‐Sottas syndrome (DSS), and congenital hypomyelination (CH). Authors of the 2006 study say: “To date, 11 mutations have been reported in HNPP patients and all of them are likely to cause a loss of function of the protein.” An earlier 2003 report suggested there were “fewer than 10 point mutations of the PMP22 gene” associated with HNPP.
CMT1B
CMT1B is the second most common subtype of CMT1. CMT1B is caused by a defect within the MPZ gene, which lies on chromosome 1. The MPZ gene produces myelin protein zero, and disruption of this gene also causes deficits within the myelin sheath. CMT1B patients have onset and symptoms similar to those of CMT1A patients, although there is a wide range of variability within CMT1B. As discussed in the article When HNPP ‘Causes Breathing Problems’, the MPZ gene is known to be associated with respiratory issues which is less common with HNPP.
CMT1E
Similar to point mutations in HNPP, instead of having a duplication of the normal PMP22 gene, CMT1E patients harbour different genetic abnormalities in the PMP22 gene.
CMTX
CMTX is caused by mutations in the gene for connexin 32, which normally codes for a protein located in myelin, the insulating sheath that surrounds nerve fibres. It has many of the same symptoms of CMT1 and CMT2, including muscle weakness and atrophy, and changes in sensation, mostly in the feet, lower legs, hands and forearms. However, because of its linkage to the X chromosome, CMTX often affects males more severely than females.
CMT2
CMT Type 2 (CMT2) is a subtype of CMT that is similar to CMT1 but is less common. It is typically autosomal dominant, but in some cases can be recessive. CMT2 is caused by direct damage to the nerve axon itself in comparison to CMT1 which results from damage to the myelin sheath insulating the axon. CMT2 is commonly referred to as “axonal” CMT.
CMT2A is the most common subtype of CMT2 and is caused by defects in the MFN2 gene. The MFN2 gene encodes for Mitofusin 2, which is a protein involved in the fusion of cellular mitochondria. Other more rare forms of CMT2 and their gene defects include:
CMT4
CMT4 is a rare subtype of CMT that is inherited in an autosomal recessive pattern. Generally, cases of CMT4 present with more severe symptoms compared to CMT1 or CMT2. In general, CMT4 is caused by defects in the myelin sheath which insulates the axon. However, other variations include:
Source: CMT USA
Asked about the similarities of Chronic inflammatory demyelinating polyneuropathy with HNPP and CMT, the symptoms that manifest seem to overlap with HNPP and CMT. However, the major difference is the fact that it is not an inherited condition, but a neurological disorder that causes progressive weakness and impaired sensory function in the legs and arms. No clear genetic predisposition or other predisposing factors for CIDP have been identified.
CIDP is thought to be caused by the immune system mistakenly attacking and damaging the myelin sheath of the peripheral nerves hence the symptoms may appear to be similar to HNPP and CMT. It is said that many people with CMT are initially diagnosed with CIDP due to these symptoms.
After that short science lesson, it’s clear to see how easily one condition can be mistakenly diagnosed for another. The symptoms tend to overlap with one another because the end result can include damage to the myelin sheath and neuropathic tendencies. As a result, it requires careful analysis by health professionals, and the necessity that all essential medical tests are undertaken to get the correct answers.
Read: Why do some HNPP results appear negative?
Read: HNPP, belief and the impact of misdiagnosis

There are days where you don’t have the time, energy, let alone the funds to help yourself. And as a result reaching out can be a lot of effort. So with that in mind, how do you socialise without leaving the house?
Read: How HNPP can cause isolation
For many people, social networks are a place for idle chatter about what they made for dinner or sharing cute pictures of their pets. But for people living with chronic diseases or disabilities, they play a more vital role.
According research conducted by the University of Warwick, chronically sick people do not have lower rates of social participation per se. However, the pattern of social participation exhibited by people affected by chronic illness varies from the one exhibited by people not affected.
“If they can break free from the anchors holding them down, people living with chronic disease who go online are finding resources that are more useful than the rest of the population.”
Susannah Fox, associate director of digital strategy – Pew Internet and American Life Project
Across all types of chronic illness the pattern of activities in which people affected by chronic illness engage in is very similar, therefore when it comes to social participation, the decisive point is whether one is affected or not by chronic illness and how it may emotionally affect people suffering from HNPP.
One of the most telling things you will find is the absence of resources devoted to help those battling with illnesses to actually make this leap. Instead, there is a wealth of literature pointing to the fact that lack of social interaction is actually a problem.
What technology is out there?
We are fortunate however, to live in an age where social networks make it easy to reach out to others. These are especially helpful when illness and pain prevent us from leaving our homes.
Many different e-social activities, including email and instant messaging, give you an opportunity to stay connected daily. It does not matter whether you are reaching out to friends, family, or online acquaintances; the important thing is that you are connecting and not struggling alone.
So without further ado, here are a few apps, gifts and devices that you can use without leaving your sofa:
https://www.instagram.com/p/BXPKgivggCu/?taken-by=twistedchronicwarrior
Obviously joining real support groups and taking part in the real world is the best way of not becoming isolated. And while it is important to have a network of people to relate to, there will be times when life requires you to be alone or when you simply want to be alone.
The point is to strike the right balance and not allow isolation to take over your life. Get out there in the real world or utilise the internet. Reach out to friends, loved ones, and acquaintances, or try and meet some new friends. You should also enjoy your alone time when it is needed.

Four weeks of having hot flushes made me wonder if I was getting my menopause 20 years too early. Or whether it could be related to medications. An even more plausible explanation could be that our internal temperature gauge may be functioning improperly with HNPP. So how sensitive are our bodies to hot and cold?
Peripheral nerves connect the brain and spinal cord to muscles and to sensory cells that detect sensations such as touch, pain, heat, and sound. Damage to the peripheral nerves can result in loss of sensation as well as wasting (atrophy) of muscles in the feet, legs, and hands.
In spite of this, the control of body temperature takes place in the central nervous system at different levels, from the spinal cord to the upper parts of the brain. According to the 1996 report The Autonomic Nervous System and Body Temperature by R. H. Johnson, the maintenance of body temperature depends upon a balance between heat loss and heat production. The mechanisms by which heat loss and production are regulated are very similar; each may be regulated in two ways:
However, instead of feeling the extremes of hot and cold due to autonomic neuropathy, studies suggest that we’re more likely to feel nothing at all. HNPP’ers on the other hand have spoken about becoming particularly sensitive to the weather.
“It’s been cold this week, I’m now sporting three pairs of socks and my feet still feel cold! I don’t think it’s to do with circulation, more misfiring nerve-endings, which just scream ‘cold’. The odd thing is though is that I’ve been getting burning sensations in my feet… burning cold.”
From Jon Leonard’s blog – My Life with HNPP
The idea of loss of sensation can be seen in PMP22 Mutation Causes Partial Loss of Function and HNPP-like Neuropathy. Both a 35-year-old man and woman complained of a “decrease in pain and temperature sensation”. The authors say in these cases “heterozygous T118M mutations mildly disrupt myelin to an extent that mild symptoms are likely to develop; homozygous mutations more severely disrupt peripheral nerve and disable patients”. But this appears only in this kind of specific mutation.
According to the 1997 study Hereditary Thermosensitive Neuropathy: An Autosomal Dominant Disorder of the Peripheral Nervous System, both Charcot Marie-Tooth disorder and HNPP had no links to this condition. HTN is also autosomal and inherited, with patients suffering from body temperatures of over 38.5 C.
The reason the authors give for it being unrelated was: “We excluded loci causing other hereditary demyelinating neuropathies, such as Charcot-Marie-Tooth disease type I (CMT type I) and hereditary neuropathy with liability to pressure palsies (HNPP), by linkage analysis; thus, HTN is not allelic to either CMT type I or to HNPP.”
https://twitter.com/tattooed_mummy/status/615985345344204800
This is reiterated in the book Peripheral Neuropathy in Childhood in which the authors Robert A. Ouvrier, J. G. Mcleod, and J. D. Pollard mention the above study adding that Hereditary sensory and motor neuropathy (HSMN) was also excluded from the research. That being said, there is very little research into this condition, with many journals relying simply on the 1997 report.
Why temperature is seen to be connected
Interestingly, the report by R. H. Johnson as mentioned above, does put a small disclaimer about body temperature being controlled by the central nervous system, saying: “Strictly speaking heat production by shivering is mediated by motor nerves but is included so that temperature regulation may be kept in proportion.”
He goes on to say that there is a combination of peripheral nerve issues as well as the central nervous system that causes problems with temperature: “The abnormalities of temperature regulation occur with failure of peripheral effector structures, alteration of function of central controlling structures or with lesions of interconnecting nerves.”
Why temperature in HNPP is seen to unrelated
This can be seen in several studies including Thermoregulation in Peripheral Nerve Injury-Induced Cold-Intolerant Rats. In 2012, scientists from the University Medical Centre Rotterdam, Netherlands, tested cold temperatures on rats that had peripheral nerve injuries. By attempting to re-warm the paws of the animals over three to nine weeks, the researchers concluded that “re-warming patterns are not altered after peripheral nerve injury in these rat models despite the fact that these animals did develop cold intolerance”.
They go on to add: “This suggests that disturbed thermoregulation may not be the prime mechanism for cold intolerance and that, other, most likely, neurological mechanisms may play a more important role.
“There is no direct correlation between cold intolerance and re-warming patterns in different peripheral nerve injury rat models.”
Peripheral nerves and thermoregulation
It’s obvious that the peripheral nerve system does play a part in feeling the differences between temperatures. In an 2009 study, nerve conduction tests were carried out on 45 Indian women between the ages of 18-25. The researchers say: “Patients with impaired circulation may have a reduced tissue temperature and additional reduction of nerve conduction velocity.” Information carried from peripheral temperature sensors tends to effect the temperature of the skin.
Authors of the 1985 research Skin-Temperature Stability between Sides of the Body reinforce this notion saying: “In normal persons, the skin temperature difference between sides of the body was only 0.24 degree +/- 0.073 degree C. In contrast, in patients with peripheral nerve injury, the temperature of the skin innervated by the damaged nerve deviated an average of 1.55 degrees C.”
It’s evident that the central nervous system, and more importantly key brain centres are mainly concerned with temperature control as well as the preoptic area and anterior hypothalamus – a portion of the brain that contains a number of small nuclei with a variety of functions.

However, scientists at the Max Delbrück Center for Molecular Medicine in Berlin, Germany, states that these hypothalamic regions harbour neurons not only detect changes in core body temperature, but are also believed to “receive and integrate input from ascending somatosensory pathways carrying information from peripheral temperature sensors”.
Despite those with HNPP clearly suffering from problems with temperature regulation, unless the connection between the autonomic nervous system is more distinctly laid out, research in this area will continually be put on the back burner. Nonetheless, there are reasons behind why we may feel cold during particularly bad weather so you’re not the only one.

Not everyone faces this problem thankfully, but for those who do, showering and generally washing can be an absolute pain with HNPP. The energy needed to stay clean is surprisingly high on the list of exhaustive tasks. From lathering, to attempting to be dexterous and flexible washing your back, it quickly becomes a tedious endeavour.
Okay, I’m exaggerating a tiny bit, but we all know that staying clean and presentable costs us much more in pain and fatigue than our healthy counterparts. I say this because the gaps between washing appear to have increased since being diagnosed with HNPP. In some cases, showers can exacerbate pain, exhaustion or vertigo and on some days, they’re just not going to happen.
Here are some hacks some may use to make showering (or bathing) with chronic illness easier. If you struggled to shower today, that’s okay. You’re not alone.
Some tips before undertaking this task:
What to think about while washing:
The biggest ‘hack’ is to remind yourself that you do have a health condition, so if you don’t have the energy to shower one day, it’s completely okay.

Dedicated to all the wonderful friends and family in the world helping those with HNPP.
Last week was particularly frustrating. After two weeks of eating TV dinners, my friends came over and cooked me my first proper meal in a while. And following the warm, fuzzy feelings of love and kindness, guilt began to set in. So why do we feel guilty about our attempts to cope with HNPP and asking for help? This may not be the first post about guilt, however, it’s an important factor to think about within relationships.
Feeling guilty comes from the notion that you’re doing something bad or wrong when you don’t meet your self expectations or when you don’t meet the expectations of others. But in being sick and in being physically limited by your illness, you’re not doing anything bad or wrong. When it comes to an illness, there is a difference between doing something and being something.
Kathy Charmaz, Professor of Sociology at Sonoma State University in California, analysed the consequences of the loss of self in the 1983 article A Fundamental Form of Suffering in the Chronically Ill. In it she says that because suffering loss of self develops out of daily life, social psychological conditions contribute to that suffering.
She states: “When ill persons attempt to return to the normal world and fail, they usually feel profound disappointment and grief for their lost self-images. As discrediting events recur, the chronically ill begin to see themselves as permanent failures and as burdens to others.”
The powerlessness of not being to ‘perform’, and therefore becoming dependent on others can be a major source of worry for some who socially identify on being independent. My friends may see me hovering around the kitchen as they cook for a reason.
Read: HNPP and the impact of chronic pain on relationships
Professor Charmaz adds: “Since becoming a burden is rooted in physical dependency and immobilization, ill persons usually recognize that their illnesses have become their major source of social identity […] Becoming a burden affirms and intensifies immobility and stands in symbolic contrast to the way these persons wish to conceive of themselves.”
Becoming ‘a burden’ means that a person no longer fulfils the obligations implicit in past relationships. Even though what constituted ‘fulfilling obligations’ may only be realised when someone is no longer able to do it, guilt and shame about burdening others usually follows. And perhaps the worst part of this sense is the feeling of uselessness to yourself and others even though those roles and obligations may never have explicitly been expressed.
What becomes obvious is that there is a heightened sense of self-concern about the person we see ourselves becoming and about valued self-images from the past which we have lost after being diagnosed with a chronic illness.
“Guilt was something that forced me to overcompensate in every aspect of my life. I was overindulging and swimming in my own guilty conscience. I thought I had a right to feel guilty because I wasn’t able to participate in physical activities with my children or because I was no longer cooking meals from scratch or that our home wasn’t impeccably clean. I spent so much time overreacting that I didn’t see the bigger picture, where my children didn’t care about any of these things – at least not in the way I did.”
Lana Barhum – Rheumatoid Connect
In the study, Professor Charmaz alludes to the fact that as a result of negatively comparing our current level of functioning to past levels and previous personal performance standards, individuals “suffer tremendous amounts of self-blame and guilt”.
Consequently, she goes on to add: “they may devote much energy to apologizing to others for their felt inadequacies. Indeed, they may apologize for their very existences because they too share the assumption that in order to be fully human, one must be able to function fully.” This may in turn result in those suffering from chronic illnesses to try and overcompensate.
https://twitter.com/lizardbonez/status/895028657521131520
How does this manifest?
If you do not have a form of chronic illness, you may be perplexed right now. There are many different sources of this guilt. These may include:
How to stave off feelings of guilt
When any of the above scenarios are in play, guilt is often self-inflicted, which is hard enough. But when family or friends add to the guilt, it makes those feelings come alive and place a much heavier burden on the chronically-ill person. For our own health it’s important to thwart these feelings and thoughts. And when in doubt, be open and honest with your friends and family and let them know how you feel.